Background:Early T-cell Precursor Acute Lymphoblastic Leukemia (ETP-ALL), an orphan disease, is a sub-type of T-Cell Acute Lymphoblastic Leukemia (T-ALL) with very poor prognosis and limited therapy options. ETP-ALL is a heterogeneous disease with many distinct genomic profiles, often with more myeloid than lymphoid characteristics. However, standard of care (SOC) drugs for acute myeloid leukemia (AML) have shown limited efficacy for ETP-ALL (PMID: 32733662, 25435716). The genomic profiles of ETP-ALL patients have more complex cytogenetics and larger numbers of genomic aberrations when compared to non-ETP-ALL (T-ALL) profiles (PMID: 22237106, 30641417).
We present an alternative multi-gene analysis approach using the Cellworks Omics Biology Model (CBM) workflow to identify unique, intersecting protein pathways in patient-specific disease profiles. The CBM predictive workflow was used to design novel personalized therapy options for an ETP-ALL representative PEER human lymphoid cell line in comparison to a T-ALL JURKAT cell line. The predicted combination therapies were then validated in a lab model.
Methods:A PEER cell line was selected to represent ETP-ALL and a JURKAT cell line was selected as a representative for non-ETP T-ALL. Next Generation Sequencing (NGS) was performed for the PEER cell line. For the JURKAT cell line, publicly available NGS whole exome sequencing from cBioPortal and Sanger, along with array CGH from Agilent, were used.
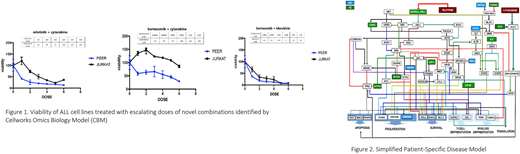
The genomic data for the PEER and JURKAT cell lines were used as inputs to the CBM to generate dynamic patient-specific disease protein network maps. Biomarkers and pathway characteristics unique to the PEER and JURKAT cell lines were identified. A digital drug library of targeted FDA-approved agents was simulated on the disease models using both single drug agents and drug combinations at varying doses. The treatment impact was assessed by quantitatively measuring drug effect on a cell growth score, which is a composite of the quantified values of cell proliferation, survival and apoptosis along with impact on the patient-specific disease biomarker score. Comparative dose response studies were run to assess IC50 differences for both cell lines. Cellworks VenturaTM predicted novel therapy combinations for the ETP-ALL representative PEER cell line, which were then prospectively validated by in vitro experiments. The same therapy options were predicted to be less effective in the T-ALL representative JURKAT cell line, which was also confirmed by in vitro studies.
Results:The CBM predicted three novel combination therapies for the ETP-ALL representative PEER cell line: nilotinib + cytarabine, bortezomib + cytarabine and bortezomib + idarubicin. All three therapies were predicted to be less effective in JURKAT cells. In vitro, PEER cells were sensitive to all 3 combinations, as predicted by the CBM; whereas, JURKAT cell lines were not sensitive to the first 2 combinations (as predicted), but were sensitive to bortezomib + idarubicin.
The CBM analysis is supported by scientific rationales for these combinations based on the genomics-driven disease characteristics of the cell-line. The reasons for drug sensitivity and resistance were determined. These combinations were then prospectively validated in vitro on both cell lines and the experimental responses matched the predicted outcomes.
Conclusion:The Cellworks Omics Biology Model integrates the multiple genomic abnormalities in a patient to identify disease network characteristics unlike other NGS analytic tools that attempt to interpret the impact of each genomic alteration in isolation. CBM identified 3 novel therapy options for ETP-ALL that were validated in vitro, similar to anecdotal experience in vivo. This predictive technology can improve clinical decision-making and identify novel treatment options.
Howard:Cellworks:Consultancy;Servier:Consultancy, Other: Speaker;EUSA Pharma:Consultancy;Sanofi:Consultancy, Other: Speaker;Boston Scientific:Consultancy.Kumar:Cellworks Research India Private Limited:Current Employment.Pampana:Cellworks Research India Private Limited:Current Employment.Ullal:Cellworks Research India Private Limited:Current Employment.Tyagi:Cellworks Research India Private Limited:Current Employment.Lala:Cellworks Research India Private Limited:Current Employment.Kumari:Cellworks Research India Private Limited:Current Employment.Joseph:Cellworks Research India Private Limited:Current Employment.Raju:Cellworks Research India Private Limited:Current Employment.Balakrishnan:Cellworks Research India Private Limited:Current Employment.Mundkur:Cellworks Group Inc.:Current Employment.Macpherson:Cellworks Group Inc.:Current Employment.Nair:Cellworks Research India Private Limited:Current Employment.Kapoor:Cellworks Research India Private Limited:Current Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal